Healthcare and Laboratory Labels Market Forecast 2026–2036: Global Market to Reach USD 11.2 Billion by 2036 at 2.7% CAGR
USA healthcare & lab labels to grow at 3.7% CAGR by 2036, driven by compliance, standardization, and UDI rules boosting demand for reliable labeling solutions.
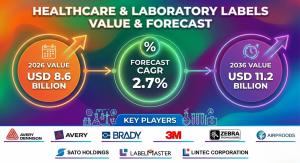
NEWARK, DE, UNITED STATES, February 23, 2026 /EINPresswire.com/ -- The global healthcare and laboratory labels market is entering a phase of compliance-driven transformation, where labeling systems are evolving from basic identification tools into critical components of patient safety, traceability, and automated data workflows. According to Future Market Insights, the market is projected to grow from USD 8.6 billion in 2026 to USD 11.2 billion by 2036, registering a compound annual growth rate (CAGR) of 2.7% over the forecast period.
This growth reflects a structural shift in procurement behavior across healthcare systems, diagnostic laboratories, pharmaceutical companies, and research institutions. Labels are increasingly specified as compliance-critical assets, supporting identity assurance, auditability, and data continuity in high-risk clinical and laboratory environments.
Key Market Highlights
• Market Value (2026): USD 8.6 Billion
• Projected Value (2036): USD 11.2 Billion
• Forecast CAGR (2026–2036): 2.7%
• Leading End User: Hospitals (40.0% share)
• Key Growth Countries: United States, United Kingdom, China, Japan, India
• Key Companies: Avery Dennison, Brady, 3M, Zebra Technologies, Sato Holdings, Labelmaster, Lintec Corporation
Discover Growth Opportunities in the Market – Get Your Sample Report Now
https://www.futuremarketinsights.com/reports/sample/rep-gb-12535
Market Overview: Labels Become Core to Patient Safety and Data Continuity
Healthcare and laboratory labels are no longer viewed as consumables. Instead, they are embedded into workflows that require consistent identification, traceability, and error mitigation. The economic impact of labeling failures ranging from clinical risk to regulatory exposure is driving demand for durable, high-performance solutions.
As healthcare systems digitize and laboratory automation expands, labels now function as both physical identifiers and digital data carriers. This dual role is positioning labeling systems as a foundational layer in patient safety frameworks and diagnostic reliability.
Compliance Standards Redefine Performance Expectations
Regulatory mandates are elevating labeling performance requirements across healthcare and laboratory environments. Frameworks such as Unique Device Identification (UDI) and direct marking requirements under 21 CFR 801.45 are increasing the cost of label failure, pushing procurement teams to prioritize durability and long-term reliability.
Labels must now withstand sterilization cycles, chemical exposure, abrasion, and repeated handling without compromising readability or adhesion. This shift is transforming labels into compliance-critical infrastructure rather than packaging components.
Persistent identification requirements are driving demand for advanced materials, including adhesive systems and face stocks validated for harsh clinical conditions and extended lifecycle use.
Automation Drives Adoption of Smart Labeling Technologies
Laboratory automation is reshaping labeling requirements, particularly in high-throughput environments where manual scanning and orientation are no longer feasible. Robotics, conveyor systems, and enclosed workflows are increasing reliance on non-line-of-sight readability.
As a result, demand is rising for RFID and advanced 2D barcode technologies that support automated identification without manual intervention. These smart labeling systems combine tag design, printer calibration, and verification tools to ensure consistent scan performance.
Automation is also increasing the strategic value of labels, positioning them as enablers of throughput optimization, traceability, and error reduction. Label failures in automated systems can disrupt workflows and introduce data integrity risks, further reinforcing the need for high-performance solutions.
Cost Volatility and Trade Policy Shape Adoption Patterns
Despite long-term momentum toward smart labeling, adoption is moderated by input cost volatility and trade policy uncertainties. RFID deployment remains sensitive to tag pricing, inlay availability, and substrate costs.
Healthcare and laboratory buyers are prioritizing smart labeling implementation in high-risk workflows, such as specimen tracking and critical device identification, while maintaining barcode-based systems in lower-risk applications.
This has resulted in hybrid labeling environments, where traditional and smart technologies coexist as cost management strategies rather than technical limitations.
End-User Segmentation: Hospitals Lead, Labs Drive Innovation
The healthcare and laboratory labels market serves diverse end users, with hospitals accounting for approximately 40% of total demand in 2026. Hospital environments present the highest concentration of risk-bearing workflows, including medication administration, specimen collection, and sterile processing.
Hospital demand is sustained by:
• The need for labels that withstand sterilization, chemical exposure, and handling stress
• Interoperability requirements across printers, scanners, and information systems
• High operational and regulatory costs associated with misidentification
Diagnostic laboratories represent the most automation-sensitive segment, where demand is concentrated on labels capable of withstanding cold storage, centrifugal forces, and high-speed application.
Pharmaceutical companies and research institutions prioritize labeling as part of quality systems, with demand focused on chain-of-identity, audit readiness, and compliance across controlled environments.
Regional Outlook: Strong Growth in Asia and Emerging Markets
While the global market is projected to grow at 2.7% CAGR, several countries are expected to outperform due to structural demand drivers:
• Japan: 4.5% CAGR, driven by advanced automation and strict compliance standards
• India: 4.4% CAGR, supported by expanding healthcare infrastructure and diagnostic capacity
• China: 4.1% CAGR, reflecting scale expansion and regulatory tightening
• United States: 3.7% CAGR, driven by compliance upgrades and automation economics
• United Kingdom: 3.5% CAGR, shaped by centralized procurement and traceability standards
Competitive Landscape: Integration and Validation Drive Leadership
The competitive landscape is defined by portfolio breadth, materials expertise, and the ability to integrate labeling systems into broader data and automation ecosystems. Leading companies such as Avery Dennison, Brady, 3M, Zebra Technologies, Sato Holdings, Labelmaster, and Lintec Corporation are competing across multiple dimensions.
Key competitive factors include:
• Performance in harsh clinical and laboratory environments
• Integration with scanning, printing, and data capture systems
• Validation, documentation, and regulatory compliance capabilities
Suppliers that reduce qualification complexity and minimize scan failure risk are expected to gain market share as healthcare and laboratory environments become more regulated and automation-intensive.
Why FMI: https://www.futuremarketinsights.com/why-fmi
Have a Look at Related Research Reports on the Packaging Domain:
Packaging Waste Recycling Market https://www.futuremarketinsights.com/reports/packaging-waste-recycling-market
Packaging Foam Market https://www.futuremarketinsights.com/reports/packaging-foam-market
Decorative Lighting Packaging Market https://www.futuremarketinsights.com/reports/decorative-lighting-packaging-market
Ceiling Light Packaging Market https://www.futuremarketinsights.com/reports/ceiling-light-packaging-market
About Future Market Insights (FMI)
Future Market Insights, Inc. (FMI) is an ESOMAR-certified, ISO 9001:2015 market research and consulting organization, trusted by Fortune 500 clients and global enterprises. With operations in the U.S., UK, India, and Dubai, FMI provides data-backed insights and strategic intelligence across 30+ industries and 1200 markets worldwide.
Sudip Saha
Future Market Insights Inc.
+1 347-918-3531
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.